Anemia with mean corpuscular volume (MCV) above the upper limit of normal (usually ≥ 100 fL) is considered macrocytic anemia. The numerous causes of macrocytic anemia can be divided into major categories (1,2) (Figure 1).
First, a reticulocyte production index should be calculated and if elevated the MCV can be above the normal range due to the large size of reticulocytes. Once high MCV is not thought to be related to reticulocytosis, the majority of macrocytic anemias can be categorized according to one of two major mechanisms: 1. Liver disease; and 2. Impairment of DNA synthesis, which includes nutritional deficiencies (folate, B12), drug effect (e.g co-trimoxazole, anti-neoplastic agents and certain anti-retroviral drugs) and “idiopathic” causes (myelodysplastic syndromes).
Mild macrocytosis can also be seen in hypothyroidism and hypoproliferative anemias such as aplastic anemia. Macrocytosis without anemia or liver disease can also be a manifestation of heavy alcohol intake.
Macrocytic anemia in liver disease is due to excess lipid deposition in the red blood cell (RBC) membrane, not impairment of DNA synthesis. Enlarged RBCs are usually round and often have a targeted appearance in liver disease; acanthocytes (spur cells) may also be present (Fig 2). In contrast, in disorders of impaired DNA synthesis, enlarged RBCs are often oval-shaped (macro-ovalocytes) (Fig 3).
Other common abnormalities seen with macrocytic anemia include hypersegmented neutrophils (eg, induced by B12 or folate deficiency), and in the case of myelodysplastic syndromes, hypogranulated neutrophils and Pelger-Huet neutrophil abnormalities.
Bonus pearl: Did you know that the MCV unit, fL, stands for femtoliters or 1/1,000,000,000,000,000 L?
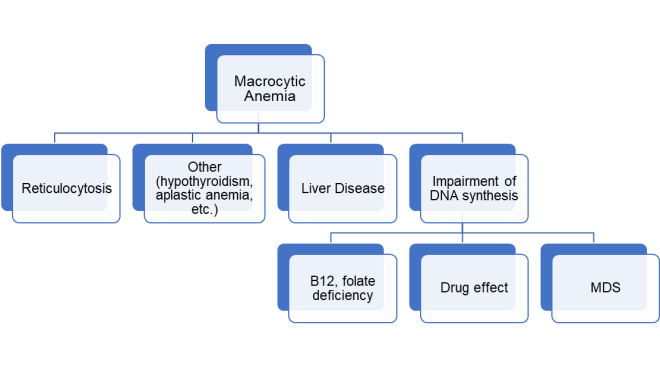
Figure 1. Major causes of macrocytic anemia. MDS: myelodysplastic syndrome.

Fig 2. Round macrocytes with targeting and abundant acanthocytes (spur cells) in a patient with hepatic cirrhosis.

Fig 3. Oval macrocytes in a patient with large granular cell leukemia and an MCV of 125 fL who received cyclophosphamide.
References
- Ward PC. Investigation of Macrocytic Anemia. Postgrad Med 1979; 65: 203-207. https://www.ncbi.nlm.nih.gov/pubmed/368738
- Green R, Dwyre DM. Evaluation of macrocytic anemias. Semin Hematol 2015; 52: 279-286. https://www.sciencedirect.com/science/article/abs/pii/S0037196315000554
Contributed by Tom Spitzer, MD, Director of Cellular Therapy and Transplantation Laboratory, Massachusetts General Hospital, Boston, MA.

